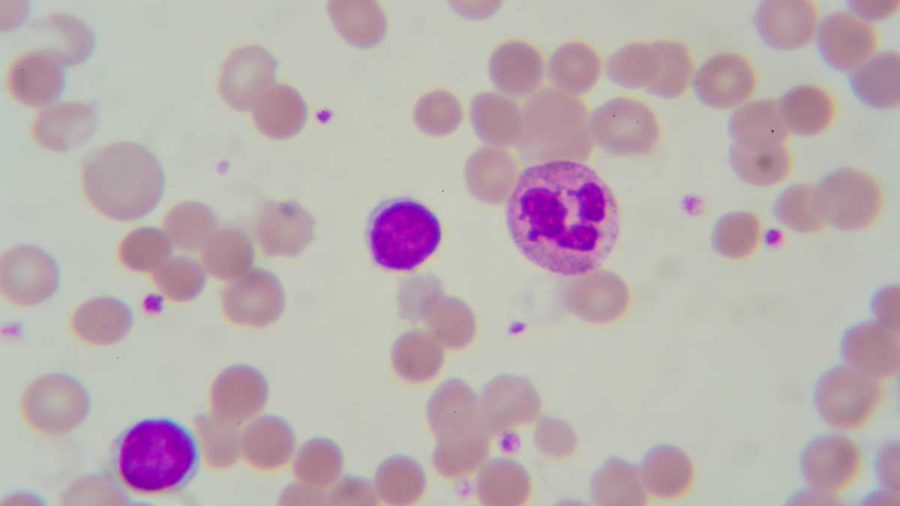
Each year, thousands of children in the United States are diagnosed with Leukemia. Over the years the advancement of technology has allowed for many breakthroughs in cancer treatments. However, the most recent breakthrough could potentially make any child with leukemia, cancer free.
On August 30, the FDA approved a gene therapy cancer treatment for childhood Leukemia. This treatment is the first gene therapy treatment to be cleared in the United States. Unlike other treatment approaches, the CAR T-cell therapy treatment doesn’t go in and replace the disease riddled genes with healthy genes. Instead, the treatment uses the patient’s’ genetically altered immune cells to fight the disease.
The treatment process beings with the extraction of white blood cells from the patient. Once the white blood cells are extracted, they are sent off to be altered so they include the new gene, that will direct the white blood cells to kill the leukemia cells. Once the gene altering process is over, the cells are injected back into the patient, so they can work their magic.
The FDA’s approval gave the go ahead on the Novartis drug Kymriah for children and young adults whose leukemia is immune to traditional cancer treatments. According to a study conducted over the summer, 80% of patients that underwent the trial treatment, went into remission. However, the treatment process is being tested for other cancerous diseases. To learn more about this new kind of leukemia treatment visit: